What is an allograft?
An allograft is any human tissue that is transplanted from one person to another. Kidneys, hearts, corneas, bone, skin and other tissues are generously donated by individuals or their families. Allograft tissues have been used in millions of surgical procedures for many years.
What is an AlloDerm dermal graft?
AlloDerm is donated allograft skin which is processed by LifeCell Corporation to remove cells, leaving only the dermis, or bottom layer of the skin, known as the basement membrane.
How is donated skin obtained?
Tissue banks regulated by the U.S. Food and Drug Administration (FDA) throughout the United States surgically remove a thin layer of skin from donors using sterile operating room techniques. The skin is placed into an antibiotic solution and shipped to LifeCell.
How long has AlloDerm been used?
Use of AlloDerm began in 1992 for burn patients and in 1994 for periodontal surgery and plastic and reconstructive surgery. Currently, more than 25,000 patients have received AlloDerm grafts.
Is it regulated by any agency?
AlloDerm is regulated by the FDA as human tissue and is indicated for transplantation to repair or replace damaged or inadequate gingival tissue.
How is AlloDerm made?
LifeCell Corporation processes the human donor skin in order to remove the cells. This process involves removing the epidermis (the top layer of skin cells) and all of the cells in the dermis. The remaining material is a collagen framework which provides strength to the skin. There are no components left to cause the rejection or inflammation that can result with unprocessed tissue transplants. Therefore, when transplanted to a patient, the AlloDerm graft gradually becomes a natural part of the patient's own tissue and performs like normal, healthy tissue thereafter.
Are AlloDerm grafts safe?
Before LifeCell receives the skin, the tissue donors undergo rigorous screening by the Tissue Bank. The donor's medical and social history and cause of death are carefully reviewed and documented. Blood samples are extensively tested by the Tissue Banks or a certified laboratory using FDA-licensed tests. The donor must be found negative for Hepatitis-B and C, HIV Types 1 and 2 antibodies (screenings for AIDS), and syphilis. As an extra safeguard, AlloDerm grafts undergo microscopic and another analytical testing, both before and after processing, to rule out pathogenic contamination and promote uniform quality.
What about HIV transmission?
Human pathogenic viruses, including HIV, require certain types of human cells to be present and alive in order for the viruses to live and grow. These viruses essentially are "parasites" of cells and cannot reproduce without them. The AlloDerm ProcessTM removes all cells, thereby removing the components necessary for survival and transmission of these viruses. Additionally, after cell removal, the AlloDerm grafts are freeze-dried. There has never been a documented case of HIV transmission from the transplant of any freeze-dried, processed tissue graft.
How is AlloDerm used?
AlloDerm is used both as a surface graft for replacing gum tissue due to periodontal disease or cancer excision, and as an implant that is inserted under the surface of the gum to correct soft tissue defects. AlloDerm has been used both as a surface graft and as an implant for almost every area of the body. Amounts of AlloDerm used in a surgery have varied from extremely small, such as half of one square inch for gum surgery, to more than 5 square feet in burn surgery.
How is AlloDerm prepared for surgery?
In surgery, the freeze-dried AlloDerm graft is rehydrated in a sterile solution. When fully rehydrated, it is very soft and pliable, feels just like your own tissue, and can be positioned or draped wherever it is needed.
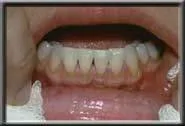
Lower recession.
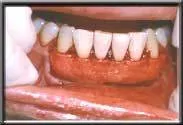
AlloDerm in place.
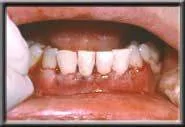
Flap sutured over AlloDerm.
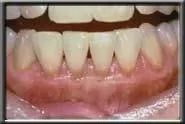
Final healing.
Why is AlloDerm preferred over other products or methods?
AlloDerm has many advantages that make it unique in comparison to other alternatives. Since it is human tissue, AlloDerm does not incite an inflammatory response or allergic reaction. Also, AlloDerm can eliminate the need to obtain tissue from the roof of your mouth, thus eliminating the need for an additional surgical site. Lastly, when used as a soft tissue graft in conjunction with bone grafts, it does not require a second surgery for removal like many synthetic products.